Organic Chemistry Laboratory II
Alkenes from Alcohols: An
E1 Elimination
Experiment Description and Background
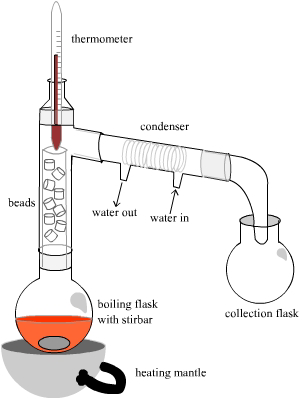
| Fractional distillation is used to separate liquids that are soluble in each other and boil within 25 °C of each other. The difference between a fractional distillation and a simple distillation is the use of a fractionating column (Vigreux or straight). The long column, filled with packing material helps to enhance the separation of the components of the mixture, ultimately enriching the distillate in the lower boiling component. In the case of this experiment, fractional distillation is used to separate the product cyclohexene (bp 83 °C) from the by-product, water (bp 100 °C). However, the fractional distillation does not provide a means to completely and thoroughly separate the two components. A residual amount of a cyclohexene-water mixture remains trapped on the packing material of the fractionating column. If the cyclohexene component of this mixture is not retrieved from the column, the yield of the reaction is severely compromised. To avoid this loss of product, a “chaser” solvent is used to “rinse off” the cyclohexene. Toluene, an aromatic hydrocarbon bp 110 °C), is used for this purpose because cyclohexene is readily soluble in this solvent, while the water is not. After most of the cyclohexene has been distilled from the reaction mixture, the toluene is added. The distillation resumes and the toluene “dissolves” the cyclohexene trapped on the packing material of the column and carries across to the collection tube. |  |
General Mechanism of the E1 Reaction
The first step of the E1 mechanism is the same as the first
step of the SN1 reaction. The bond between the leaving group
and the sp3 carbon bonded to the leaving group breaks to
generate
a carbocation. An adjacent sp3 carbon then
gives
up electrons from one of its C-H bonds to form a new pi bond between
the
carbocation carbon and the adjacent carbon. For diene formation,
the mechanism occurs so that one pi bond is completely formed and then
the second pi bond is formed in the two step sequence.
| Alkenes
|
Dienes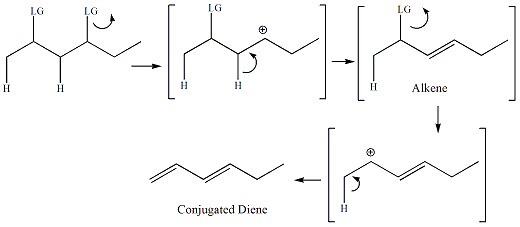 |
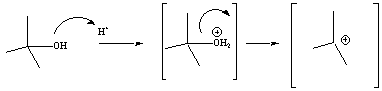
For alcohols, the OH group must first be activated with acid before
it
will become a good leaving group (LG). This is frequently done
using a strong acid (catalyst) with a conjugate base that is not
nucleophilic. Sulfuric and phosphoric acids are good activators
for E1 reactions. Hydrohalic acids (i.e., HX) are not
typically used for elimination reactions because the resulting halide
ion (conjugate base) may serve as a
nucleophile. As a result products of SN1 reactions
will be generated along with the E1 products.
Kinetically-Controlled
Reactions
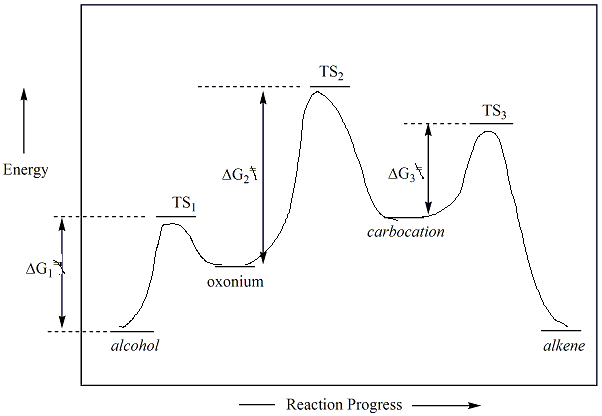
For kinetically controlled reactions, the rate determining step of this
reaction is formation of the
carbocation,
and the alkene product derived from the most stable carbocation
will be generated as the major product. A
reaction
energy diagram showing a general E1 reaction for an alkene
is given
below.
Because E1 reactions involve a carbocation, and because the
stability of the carbocation will dictate what the activation energy of
the rate determining step of the reaction is, alcohols that generate
more
stable carbocations, (i.e., tertiary) will react faster than less
stable
carbocations (secondary or primary). The rate determining step
(slowest step) in the reaction energy diagram shown is step 2
(conversion of the oxonium to the carbocation). This step has the
highest activation energy and highest energy transition state.
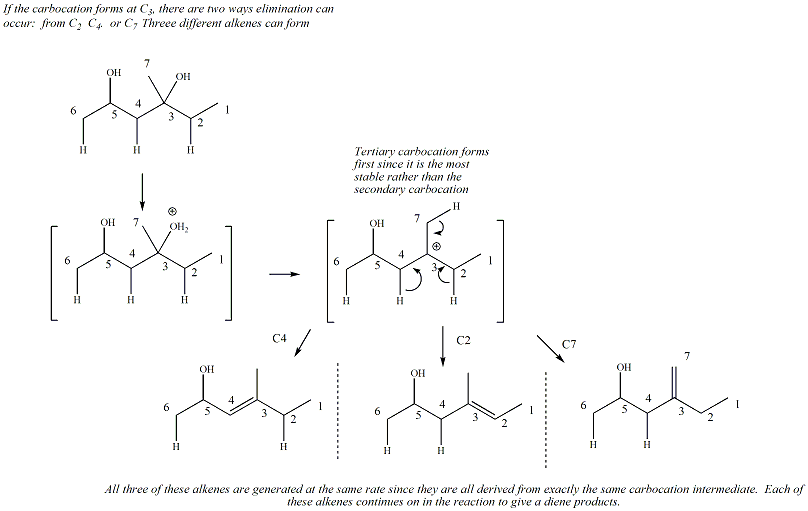
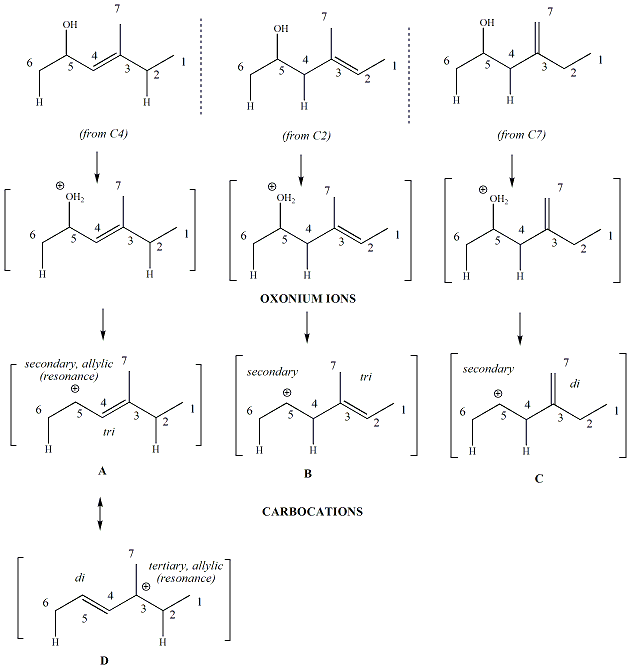
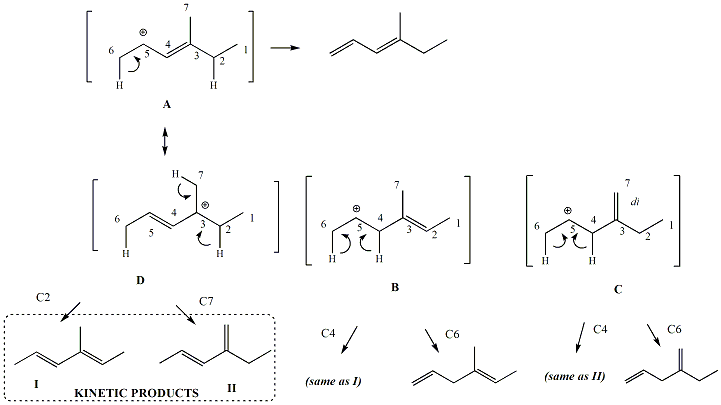
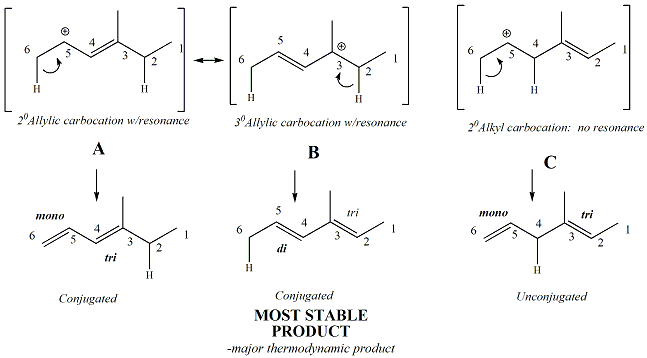
For dienes where there are two leaving
groups, the alkene that forms first under kinetic conditions will be
the alkene derived from the most stable carbocation. An example
is given below.
In this experiment, cyclohexene, an alkene, is prepared from cyclohexanol, an alcohol. Each of these compounds have unique functional groups and should therefore be easily distinguished from one another by IR spectroscopic analysis. Most characteristically, the starting material, cyclohexanol, will have a broad absorbance in the range between 3200- 3600 cm-1 corresponding to its alcohol O-H bond. This absorbance should be absent in the IR spectrum of the cyclohexene product. Some caution must be exercised however, since specific reaction conditions and by-products of the reaction must be taken into account before interpreting the IR spectra. Water is a major by-product of this reaction and as discussed above, may very likely contaminate the product, even after a fractional distillation is done. If water does in fact contaminate the product, an absorbance at 3200-3600cm-1 may appear in the IR spectrum of the product due to the O-H bond of water. Alternatively, an absorbance in this region in the IR spectrum of the product might also be interpreted as contamination by unreacted cyclohexanol. Consideration of the reaction procedure/conditions should allow you to rule this out. Why?
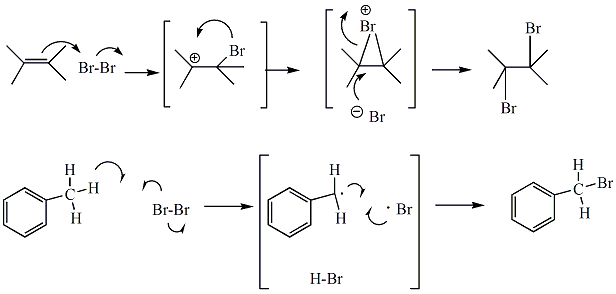
Reaction of bromine with alkenes or alkynes occurs through an electrophilic addition mechanism to generate dihaloalkanes. The brown colored bromine reagent becomes incorporated into the organic hydrocarbon, producing a colorless product. If the bromine reagent is used in excess, or if no reaction occurs, the solution will remain brownish in color. Carbon tetrachloride (CCl4) serves as solvent. Cyclohexene, an alkene, would be expected to react readily with Br2 in CCl4 to form the dihaloalkane, 1,2 -dibromocyclohexane. Bromine is a dark brown liquid, and even solutions of bromine (such as Br2 in CCl4) are brown, light brown or yellow, depending on the concentration. Dihaloalkanes, are colorless compounds. When Br2 in CCl4 reacts with an alkene, the colored bromine becomes incorporated into the colorless organic dihaloalkane, and the reaction solution goes from brown (or yellow) to colorless. Br2 in CCl4 does not react with alcohols, and the solution of Br2 in CCl4 would be expected to remain brown, light brown or yellow upon reaction with cyclohexanol. Bromine may also react with benzylic atoms through a radical halogenation mechanism. Even without a specific initiator, products from benzylic broination are often detected in this test. In the reaction of bromine with toluene, the pi bonds of the benzene ring do not react, however, the benzylic atom of toluene will be brominated in this reaction.
Jones
Oxidation Test
Jones reagent is a powerful oxidizing agent (CrO3/H2SO4)
that
is used primarily to convert primary alcohols to carboxylic acids, and
secondary alcohols to ketones. Tertiary alcohols do not react
with
Jones reagent. The carbon atom directly bonded to the OH group of
the alcohol becomes oxidized (increases its bonds to oxygen and
decreases
its bonds to hydrogen) and the chromium (VI) atom of the Jones reagent
is reduced to Cr3+.
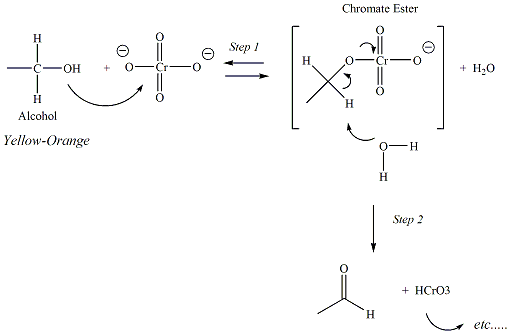
Chromium trioxide is an orange-colored or
red-orange reagent
which upon reduction
turns green or blue-green. Over the course of the organic
oxidation, the Cr is reduced
to Cr (III). The first two steps of the
reaction mechanism help to explain why tertiary alcohols do not undergo
oxidation with the Jones reagent. In
step 2, water reacts with a proton of the chromate ester that is bonded
to the
carbon atom of the former alcohol functional group.
Tertiary alcohols do not have a hydrogen atom
bonded to this carbon, therefore the reaction could not proceed beyond
this
point